|
Like lanthanides, they are radioactive, are silver and soft. Īctinides are also excluded from the main body of the periodic table but are also transition metals. They react quite aggressively with oxygen to form an oxide coating and are also radioactive. Lanthanides are found separate from the main body of the periodic table. They have low densities as well as low melting points. The next part of the periodic table is the transition metals from group 3 to 12. They also have similar reactivity with Group 1 being much more reactive than Group 2. Hydrogen is unique in that it is generally placed in group 1, but it is not a. Group 1 alkali metals, Group 2 Alkaline earth metals are both named as such as often they form alkalis. The alkali metals are lithium, sodium, potassium, rubidium, cesium, and francium. Properties commonly found in nonmetals are poor conductors of heat, brittle,Īlkali are soft metals found in group 1 that react aggressively because they have one electron in their outer shell and once they get rid of that one electron, they will obtain a full outer shell. Metals are predominantly located on the left of the periodic table. Nonmetals are found on the right side of the staircase. 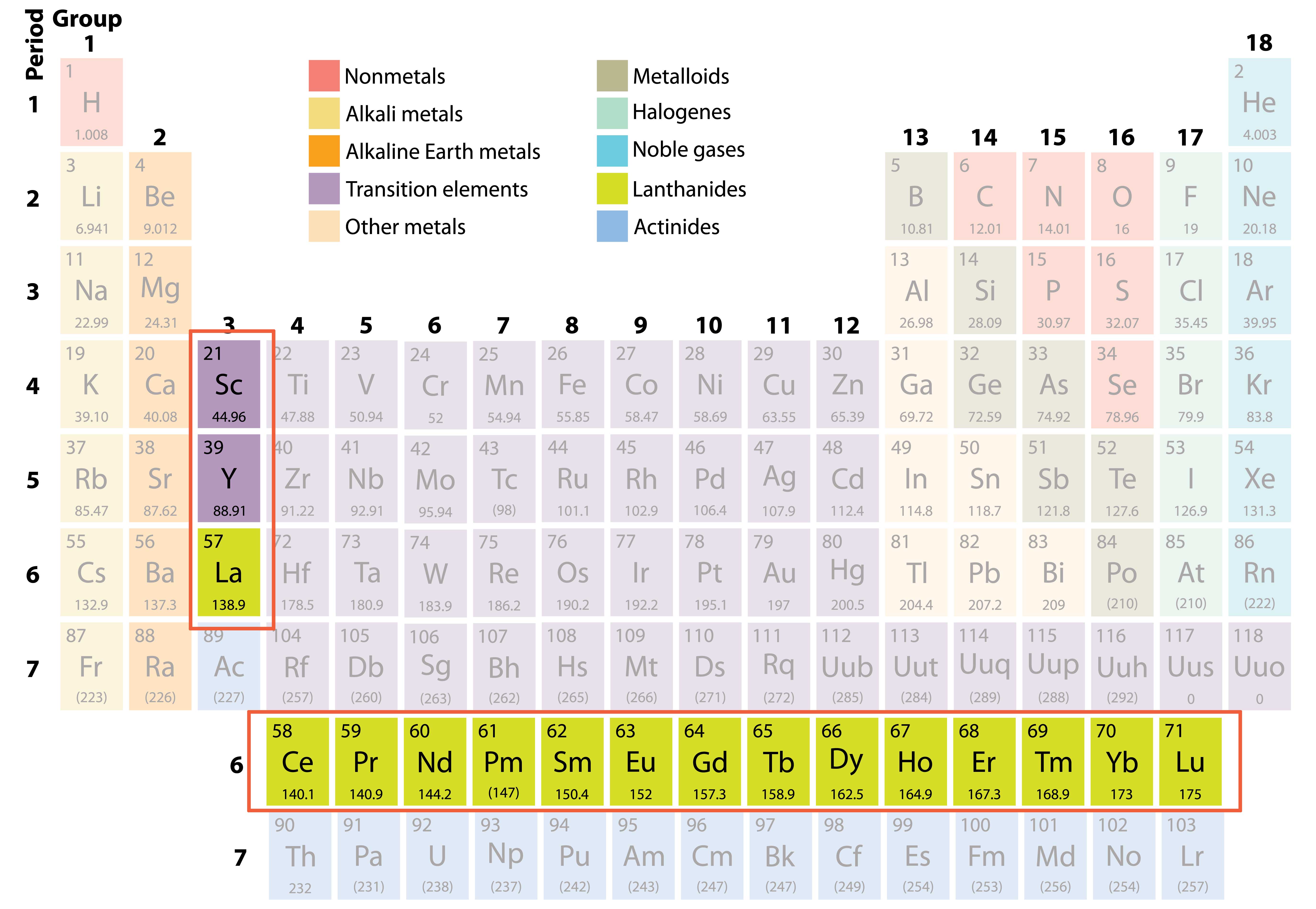
These elements are found underneath the border of the staircase. Properties usually found in metals are that they are malleable, ductile, conductors of electricity and heat and most of the time, solid. Metals are on the left side of the staircase line. They have seven electrons in their outer shell which makes them very reactive and unstable because they only need one more electron to obtain a full outer shell (8 electrons). These gases are stable elements and do not react because they have full outer shells. Properties that are commonly found in transition metals are: good conductors of heat and electricity, they are malleable (can be hit into a particular shape), usually are hard and have high densities, have high melting points. 
The section between group 2 and 3 is where the transition metals are.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
